SPERMATOGONIA STEM CELLS: A FOUNTAINHEAD OF RHINOCEROS SPERMATOZOA?
At the beginning of 2017, I moved to California to work with the Reproductive Sciences team at the San Diego Zoo Institute for Conservation Research and continue my research passion of applying assisted reproductive technologies for the preservation of endangered species. In this new program, the species to preserve were not agile predators, like the wild felids I had worked with, but they are placid grazers- the northern white rhinoceros.
The size of these animals did not undermine my purpose, but the possibility of losing them from the face of the earth encouraged me to explore which technology could be used to help save these gentle giants from extinction. To determine which technology would be the most appropriate, the first step was to determine which tissue was available. The entire population consists of three northern white rhinoceros (one male and two females) at the Ol Pejeta Conservancy in Kenya.
Fortunately, when a northern white rhinoceros male (Angalifu) living at the San Diego Zoo Safari Park died in December 2014 at 42 years of age, testicular tissue was cryopreserved and stored at the San Diego Frozen Zoo.
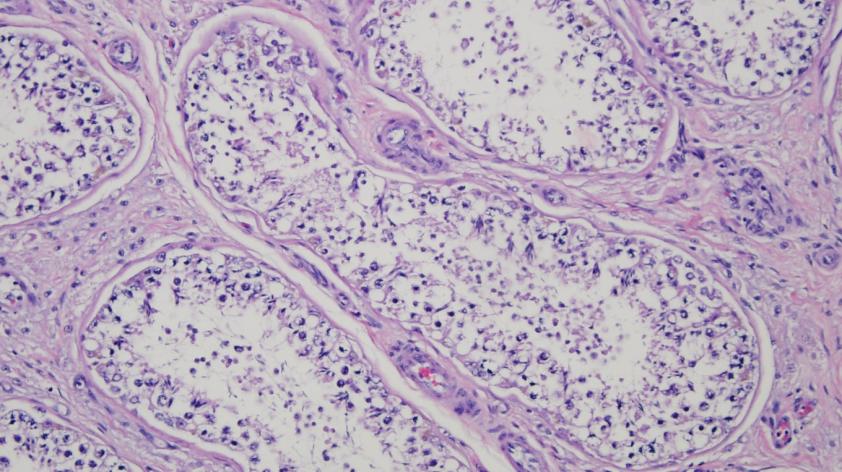
Testicular tissue contains spermatogonial stem cells, which have the unique capability of self-renewal and are responsible for the perpetual production of spermatozoa. Thus, frozen northern white rhinoceros testicular tissue potentially contains spermatogonial stem cells that could be a source of spermatozoa.
You may be wondering how these stem cells could possibly produce spermatozoa in a laboratory dish. Well, scientists are already producing sperm of wild mammalian species by isolating spermatogonial stem cells (from the testes of both live and dead animals) and transplanting them to the testis of another species (xenogenic transplantation).
In fact, in recent studies spermatogonial stem cells of ocelot (Leopardus pardalis) and lion (Panthera leo) were isolated from testicular tissue, labeled with a fluorescence dye, which allows identification of the cells for more than three months, and transplanted into the testes of domestic cats. The results are promising because both ocelot and lion spermatogonial stem cells were present in testicular tissue of the domestic cat recipient several weeks after being transplanted, and ocelot spermatozoa were obtained.
Unfortunately, due to legal restrictions and the lack of female ocelots to inseminate, scientists were unable to test the ability of ocelot spermatozoa to produce offspring. Nonetheless, other scientists have successfully produced functional sperm after allogenic transplantation (to the same species) of rhesus macaque spermatogonial stem cells.
While transplantation of spermatogonial stem cells of white rhinoceros is not presently possible, I have initiated studies to use the domestic horse (our animal model) to restore the spermatogenesis process in vitro. In addition, we are evaluating the possibility of using the horse as a recipient of white rhinoceros spermatogonial stem cells to produce functional rhinoceros spermatozoa.
By using the same markers that detected spermatogonial stem cells in the domestic horse, we have successfully demonstrated that the cryopreserved testicular tissue of the northern white rhinoceros contains spermatogonial stem cells. Our preliminary results were presented at the International Embryo Technology Society conference in Bangkok, Thailand.
We are optimistic that these exciting findings indicate that spermatogonial stem cells from Angalifu’s frozen tissue can become a fountainhead of sperm for in vitro fertilization. Then, someday, perhaps the production of his first offspring.













